On Tuesday, the Federal Trade Commission released its report on Paragraph IV litigation settlements filed in Fiscal Year 2010 (Oct. 2009 through Sept. 2010). The Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) requires pharmaceutical companies to file certain agreements, including settlements of Paragraph IV litigation, with the FTC and Department of Justice. Each year, the FTC releases a report on the settlement agreements filed in the previous fiscal year.
The FTC's report for FY 2010 states that the FTC "received 113 final resolutions of patent disputes between a brand and a generic" during the year, 31 of which "contain both compensation to the generic manufacturer and a restriction on the generic manufacturer's ability to market its product." The FTC refers to such agreements as "pay-for-delay" settlements. In a press release announcing the report, the FTC noted that these 31 agreements represent a 60 percent increase in pay-for-delay settlements over FY 2009. On a percentage basis, however, the number of such settlements remained the same in 2010 as it was in 2009.
I returned yesterday from the ACI Paragraph IV Disputes conference in New York, where Michael Kades of the FTC gave a presentation on pay-for-delay settlements. Mr. Kades noted that the increasing prevalence of such agreements since 2003,
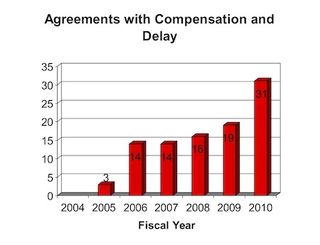 when the 6th Circuit declared a pay-for-delay settlement to be illegal in In re Cardizem, corresponds with the approval of such agreements by the 11th Circuit in 2005 (FTC v. Schering-Plough); the 2nd Circuit in 2006 (In re Tamoxifen); and the Federal Circuit in 2008 (In re Ciprofloxacin). Mr. Kades further indicated that the FTC continues to vigorously pursue antitrust cases against pay-for-delay settlements in other circuit courts, and hopes that ultimately the Supreme Court will resolve the issue or Congress will outlaw such settlements.
when the 6th Circuit declared a pay-for-delay settlement to be illegal in In re Cardizem, corresponds with the approval of such agreements by the 11th Circuit in 2005 (FTC v. Schering-Plough); the 2nd Circuit in 2006 (In re Tamoxifen); and the Federal Circuit in 2008 (In re Ciprofloxacin). Mr. Kades further indicated that the FTC continues to vigorously pursue antitrust cases against pay-for-delay settlements in other circuit courts, and hopes that ultimately the Supreme Court will resolve the issue or Congress will outlaw such settlements.
RELATED READING:

Leave a comment