Bristol-Myers Squibb v. Teva Pharms., No. 10-805-CJB (D. Del.)
Today, in the District of Delaware, Judge Burke issued a 171-page opinion holding that claim 8 of U.S. Patent No. 5,206,244, covering the entecavir molecule, is invalid as obvious. Entecavir is the active ingredient in Baraclude tablets, which are indicated for the treatment of chronic hepatitis B virus (HBV) infection. The 30-month stay of final approval of Teva's ANDA was set to expire tomorrow.
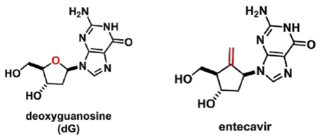
Entecavir is a carbocyclic nucleoside analog that mimics the naturally-occurring nucleoside 2'-deoxyguanosine (dG):
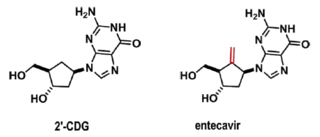
Teva argued successfully that one of ordinary skill in the art seeking to make an anti-HBV compound at the time of the invention (1990) would have selected 2'-CDG, another prior art carbocyclic nucleoside analog, as a "lead compound" and would have modified 2'-CDG at the 5' position by adding an exocyclic methylene:
Judge Burke devoted 140 pages of his opinion to findings and conclusions that entecavir would have been obvious in light of the prior art. The court held that "the prior art clearly would have directed the skilled artisan to select 2'-CDG, a carbocyclic analog, as a lead compound for further development efforts," finding that "in direct contrast to BMS's arguments in its briefing, the testimony of its own expert at trial repeatedly and conclusively established that researchers were, in fact, treating and using 2'-CDG as a lead compound in the relevant time period." Similarly, in holding that a person of ordinary skill would have had a reason to modify 2'-CDG by adding a carbon atom at the 5' position, the court found "that the powerful admissions by BMS's expert severely undercut BMS's arguments to the contrary regarding motivation to combine." The court likewise found BMS's arguments regarding reasonable expectation of success unpersuasive, stating, "notably, BMS's opening brief fails to address this factor of the prima facie case in any great detail."
Before concluding that claim 8 of the '244 patent is invalid as obvious, the court examined the objective evidence of nonobviousness, observing that "all evidence relevant to obviousness or nonobviousness [must] be considered, and be considered collectively." First, the court, "following the direction of the Federal Circuit and this Court's case law, [found] that Teva's choice to copy entecavir, while not irrelevant to the obviousness analysis, does not amount to compelling evidence of nonobviousness here." Next, the court found "that Baraclude has been a commercial success, though a less dynamic one than BMS asserts in its briefs." The court then analyzed the testimony of a BMS expert, Dr. Gish, relating to skepticism of those in the art, but found that "in the absence of additional corroborating evidence . . . [Dr. Gish's] verbal claim of skepticism is simply not enough to weigh in BMS's favor as to this factor regarding nonobviousness." The court similarly found evidence of failure of others to be weak.
The court ultimately concluded:
Teva has made out a strong prima facie case of obviousness. The evidence that it put forward at trial in that regard was multi-faceted and compelling. In addition, as to almost every significant portion of the prima facie case, Teva's position was not only bolstered by the opinion of its expert, Dr. Heathcock, but also by the testimony of BMS's expert, Dr. Schneller. On cross-examination, Dr. Schneller was forced to concede the accuracy of many significant points that Teva sought to assert as to that prima facie case. The force of this evidence was clear, and it was convincing.
As noted above, the evidence as to objective considerations was mixed. Some of those considerations redounded to the benefit of BMS's position as to nonobviousness to a degree, but as to a number of other considerations, the impact of that evidence was not particularly compelling. The totality of that evidence did not strongly persuade the Court as to entecavir's nonobviousness.
In addition to obviousness, Teva argued that the '244 patent is unenforceable due to inequitable conduct. Teva based its inequitable conduct allegation on the fact that prior art disclosing 2'-CDG was not cited to the PTO during prosecution (which, incidentally, strengthened Teva's obviousness position). Here, the court sided with BMS, finding that Teva did not demonstrate "that the single most reasonable inference in this case is that [BMS] had the specific intent to deceive the PTO, nor that the evidence requires a finding of deceitful intent in light of all of the circumstances."
Though successful in the district court, it is unclear when Teva will begin marketing its generic entecavir tablets, as Teva's ANDA has apparently not yet received tentative approval. In addition, according to the Orange Book, BMS holds a three-year exclusivity on Baraclude that expires in October 2013 and another three-year exclusivity that expires in October 2015. Meanwhile, BMS will very likely appeal today's decision to the Federal Circuit.

Leave a comment